STUART, FLORIDA November 11th, 2019: Stuart Therapeutics, Inc. (STUART), an innovative pre-clinical therapeutic development company, today announced that it has successfully completed in vitro pro-neuronal growth experiments utilizing the company’s PolyColTM (PolyCol) peptide. PolyCol demonstrated a strong propensity to encourage neurite outgrowth from dorsal root ganglia nerve cells in a model designed to mirror damage to the extracellular matrix as found in many diseases of the retina and optic nerve. These experiments, which could have far-reaching implications for regenerative medicine, are the latest in a series of tests conducted by STUART to advance the use of PolyCol as a unique therapy for challenging ophthalmic indications. STUART believes that PolyCol may be used as a treatment to promote neuronal survival, addressing the degeneration that is associated with diseases of the retina and optic nerve, including glaucoma and macular degeneration.
PolyCol’s unique mechanism of action, healing damaged supportive collagen membranes, creates a new and exciting means of dealing with nerve cell damage in disease and trauma indications. The technology has the potential to provide significant improvement for glaucoma sufferers. Glaucoma is a chronic disease affecting an estimated 76 million people worldwide. Current therapies for glaucoma impact the disease indirectly by manipulating intra-ocular pressure. These therapies fail to repair pre-existing damage, and may not work at all in a subset of the patient population.
The experiments first demonstrated the behavior of central nervous system cells (dorsal root ganglia) seeded on a healthy collagen bed. A negative control was established using matrix metalloproteinase-1 (MMP-1) to damage the collagen substrate prior to seeding the nerve cells. Finally, several variants of PolyCol were similarly applied to MMP-1 damaged collagen substrates, each followed by nerve cell application. Dendritic field area and neurite outgrowth length were measured (n was 8 for these experiments).
The PolyCol treated plates showed accelerated healing and recovery of the nerve cells:
- Dendritic field area and neurite length exceeded the negative control by 100% (p <002).
- The neurite length for this PolyCol variant exceeded that of the positive control by 70% (p < 02).
- All PolyCol variants showed benefit in either dendritic field area, neurite outgrowth length, or
- A key finding of these experiments was that nerve cell recovery in a healthy basement membrane or interstitial support structure environment is optimal
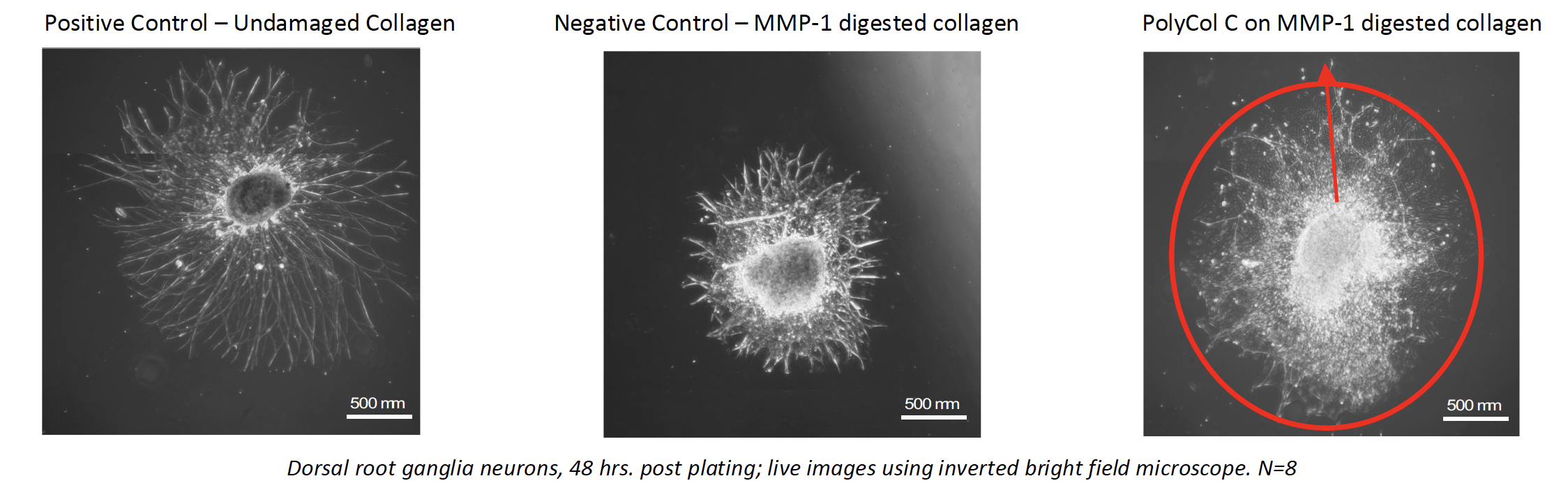
The PolyCol treated collagen substrates showed extremely rapid recovery, consistent with Stuart’s other in vivo and in vitro experimental results on ocular surface models for dry eye disease and retinal models for dry age-related macular degeneration.
“These results are extremely exciting for the Stuart Therapeutics team,” said Eric Schlumpf, President & Chief Executive Officer. “The research on nerve recovery in the eye has previously focused on molecular pathway modifiers (growth factors) or gene therapy approaches, the results of which have been dissatisfying. We see the use of PolyCol as a new, bioengineering oriented approach to repair and recovery, attacking the problem from a completely new direction.”
“As an ophthalmologist who has been frustrated for many years with the available therapies for many chronic ophthalmic disorders, the results of the studies using PolyCol have been exciting”, said Bob Baratta, MD Chief Medical Officer and Chairman of STUART. “The simplicity of the basic chemistry plus the behavior of PolyCol, having now been favorably subjected to chemical and mechanical stability testing, gives STUART strong prospects for new therapies that will stem from its research and findings to date.”
PolyCol is a collagen mimetic peptide platform. STUART controls the worldwide rights for this technology in ophthalmology therapeutics. Its form is that of a stable fractional single strand of human collagen. It acts as a direct reparative to damaged collagen in situ, and is directly applicable where disease or trauma impacts important cells and the underlying collagen membranes or structures. It repairs damaged collagen, thus improving the recovery and repair of the cellular structures dependent upon that collagen.
About STUART
Stuart Therapeutics’ mission is to offer unique solutions for difficult-to-treat diseases of the eye. STUART is focused on the research and development of ophthalmic therapeutic products based on the company’s proprietary PolyCol Collagen Mimetic Peptide platform. STUART’s first drug in development is ST-100, targeting Dry Eye Disease (DED) and other
ocular surface indications. In pre-clinical proof of concept testing, ST-100 has demonstrated accelerated repair of the epithelial layer, through binding of ST-100 to exposed sub-epithelial collagen layers. Complete repair of these tissues and the epithelial layer occurred within approximately 24 hours. ST-100 is expected to be broadly applicable to the patient population and should have therapeutic applications in patients regardless of the underlying cause of their DED. Other areas of opportunity for PolyCol include dry age related macular degeneration, for which Stuart has also developed positive in vitro results.
For more information, please visit www.StuartTherapeutics.com.
















